What is water quality? Eight key characteristics
Understanding water quality in freshwater ecosystems
Water Quality is a way of understanding the chemical, physical, and biological characteristics of a water sample to figure out how healthy it is for drinking, swimming, or supporting an ecosystem.
We rely on our waters for supplying our taps, as well as for supporting our ecosystems. Poor water quality can create health risks for people, as well as the ecosystem at large. To properly maintain water quality, we have to regularly measure each characteristic to understand its baseline — the natural resting point of our ecosystem. By regularly monitoring, we can notice trends and react early when an issue arises.
To measure water quality, testers take a small water sample and measure individual values for multiple important characteristics. These can be different depending on who is taking the measurement and why! However, there are eight key important characteristics of a water quality measurement: temperature, clarity, conductivity, pH, alkalinity, chlorine, hardness, and dissolved oxygen.
Some people think measurements are only taken by scientists, but many water quality measurements are taken by cottagers, boaters and fishers, students, and community members taking action! We can equip you with everything you need to get testing.
In this article, we will briefly explore each, as well as find out the tools and resources you need to take action!
All the tools you need are available in our flagship testkits
And make sure you browse the rest of our shop for additional tools. We will be sharing some helpful favourites below!
-
Compact Freshwater Testkit$155.00
Water temperature
Why is water temperature important?
Water temperature is a key feature of any water body. It changes the water’s density, the water’s ability to support life, as well as its ability to absorb gases (like CO₂), and nutrients.
Increases in water temperature can cause some chemicals to become “soluble”: think how quickly salt dissolves in hot water versus cold water. Algae blooms and other vegetation can also grow more quickly in warmer water. When these blooms decompose, they reduce the total amount of dissolved oxygen in the water, making it difficult for fish and other aquatic critters to breath.
How do you measure water temperature?
Water temperature is measured in degrees with a digital or analog thermometer. Make sure the thermometer is submerged at least 10 cm from the surface of the water. Hold under the water for 5 minutes. You’ll know it’s ready when the temperature has remained the same for at least 30 seconds.
Tools to measure water temperature
Water clarity
Why is water clarity important?
Water clarity is affected by both natural and human factors. When you test, be sure to note if you’ve had a recent rainfall or if you’ve noticed unusual events near the test site.
Baseline testing is important as each body of water will have unique factors contributing to water clarity. Some very healthy lakes will have very low readings if they are naturally muddy or coloured. Recording in the same place over time will give you a more accurate picture of what is normal for your body of water.
How do you measure water clarity?
Water clarity is measured with a secchi disk, a 20cm disk that is lowered into the water until it is no longer visible. Recording the depth at which the disc “disappears” gives an indication of how far sunlight can penetrate the water column.
Tools to measure water clarity
-
Secchi disk$80.00
Conductivity of freshwater
Why is conductivity important for water quality?
Conductivity is how we measure the ionic content (such as chloride, nitrate, sulfate, sodium, magnesium, calcium, or iron) in a body of water by measuring the water’s ability to conduct electricity.
We can use conductivity as an early warning system for potential problems that warrant further testing. If we see a variance (either lower or higher) from our baseline readings, we know something may have gone wrong. While lab testing is the usually the only way to determine what is causing the reading, higher conductivity readings could indicate a pollution event has taken place.
How do you measure conductivity in freshwater?
Conductivity in freshwater is measured in microsiemens/centimetre (µS/cm) using a digital probe that sends a small electrical pulse through the water. In freshwater lakes and streams, conductivity is usually between 100 – 1000 µS/cm.
Tools to measure conductivity in freshwater
-
Conductivity meter$70.00
pH of freshwater
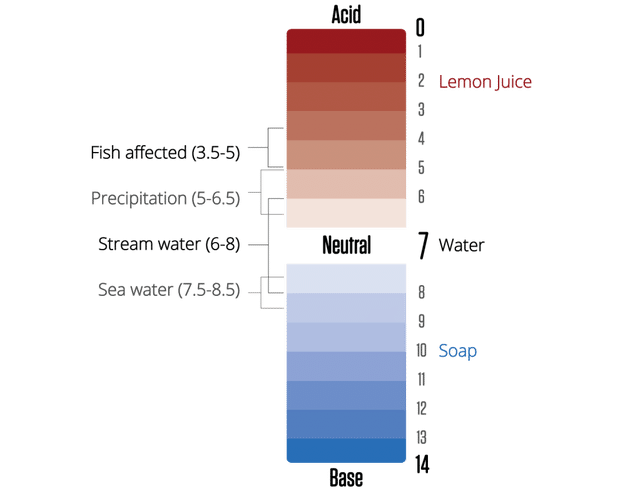
Why is pH important for water quality?
pH stands for “potential for Hydrogen”. It is the measure of the acidity or alkalinity of water soluble substances.
pH sets up the conditions for how easy it is for nutrients to be available and how easily things like heavy metals (toxicity for aquatic life) can dissolve in the water. Rivers and lakes generally range between 5 (acidic) and 9 (basic) on the pH scale. Whereas ocean water averages closer to 8.2 (slightly basic). Low pH can reduce how many fish eggs hatch and can make life difficult for fish and macroinvertebrates (the backbone of our water ecosystems).
How do you measure pH in freshwater?
pH is measured on a logarithmic scale from 0-14, with 7 indicating neutral water. There are many tools for measuring pH, from expensive digital probes to easy to use test strips. We use test strips for pH. We’ve compared 15 types of test strips, and those made by Taylor have been shown to be the most accurate.
For those looking for more accuracy, inexpensive pen-style meters have proven to be very accurate, but they need to be calibrated at least once a week. We’ve tried this one and it works well (however, you need to be careful not to dunk it too far in the water).
Tools to measure pH in freshwater
-
Teststrips$17.50
Dissolved oxygen in freshwater
Why is dissolved oxygen important for water quality?
Dissolved oxygen is the amount of gaseous oxygen dissolved in the water. Oxygen gets into the water in various ways, including absorbing it from the atmosphere, by rapid movement of the water, or as a product of photosynthesis. Oxygen is what gives the water life! Fish and plants depend on certain levels of oxygen to survive.
How do you measure dissolved oxygen in freshwater?
Dissolved oxygen is measured as a concentration, in units of milligrams per litre (mg/L).
We use the Chemetrics dissolved oxygen testkit because it is the most user friendly to conduct (no measuring chemicals) and easier to interpret than other tests we’ve tried. This device has been compared to professional probes with very good results. Full study by Carleton University to compare our kits to professional probes
The inexpensive probes that we’ve tried were finicky and inaccurate (even with proper calibration). Professional probes will give you more fine-grain results, but they’re thousands of dollars and require calibration really often.
Tools to measure dissolved oxygen in freshwater
-
Dissolved Oxygen Testkit$135.00
-
Dissolved oxygen replacement$90.00
Chlorine and freshwater hardness
Why is measuring hardness and chlorine important for water quality?
Hardness is related to alkalinity as they both measure calcium carbonate content. Hardness also measures other metals in the water that do not necessarily neutralize acid. It is one of the most common elements of testing water quality. Calcium in particular is a very important part of many of the biological processes in fish.
How do you measure hardness and chlorine in freshwater?
For this test we use Taylor Pool and Spa Test Strips. Find out why we use this brand.
Pro-Tip: If the colour on the test strip appears in the middle of two colours on the bottle, you can record the absolute middle (for example, if the colour is somewhere between 7.2 and 7.8 the recorded have a value of 7.5).
Tools to measure hardness and chlorine in freshwater
-
Teststrips$17.50
How to test the water quality of tap and well water
To measure the water quality of tap or well water, you will need to look at some additional parameters to check for metals, contaminants, and other substances that may be harmful to human health. Our kits are curated for monitoring freshwater systems, but we have a partnership with TapScore, a lab with the best drinking water test kits available, to make this experience as easy as possible for you.





